Analisys of Quality of Different Types of Honey from Various Sources

Abstract
Heavy metals pollution affects the quality of productions, as well as the qualities of the atmosphere and waters. So, the presence of metals in honey may affect the health of human as a consumer.
Analysis of the metals in honey helps its quality control as a food product. The aim of this study was to determine the concentrations of some heavy metals in some honey samples harvested from different locations in Romania using Atomic Absorbtion Spectrometer (AAS). Honey produced in different regions of Romania do not completely lack heavy metals but they are at satisfactory level and good quality for human consumption excepted the samples collected from Ploiesti, a zone with high industrial activity. The analysis shows us that in the rapes honey samples (0.15μg/g) and multiflora honey samples (0.17μg/g) the concentration maximum permitted of lead was exceeded.
Table of contents
1. Introduction
2. Materials and Methods
3. Results and discussion
4. Not detected
5. Conclusions
1. Introduction
Honey is used for nutritional, medicinal and industrial purposes and it is an important product in the international market. Honey has a superior nutritional value due to the complex composition.
Natural honey is a sticky and viscous solution with a high content of carbohydrate (mainly glucose and fructose) near water, protein, ash and minor quantities of amino acids, enzymes and vitamins as well as other substances like phenolic antioxidants [1, 2, 3]. The precise chemical composition and physical properties of natural honeys differ according to the plant species on which the bees forage.
Differences in climatic conditions and vegetation are also important factors that can affect the various properties of honey. The biochemical properties of honey and its quality are related to honey maturity, climatic conditions, production methods, processing and storage conditions, as well as the nectar source of the honey.
Metal concentrations in different honey types depend largely on the elemental composition of flowers and may come from external sources such as industrial smelter pollution, industrial unit emissions, and improper procedures during honey processing and maintenance stages. Also, the origin of metals in honey can be agrochemicals such as organic mercury, cadmium-containing fertilizers and arsenic-based pesticides.
The amount of metal in the plant increases with the amount of metal in the soil. Also, in regions where there is a significant amount of lead in the air, it may land on the plant and adhere to the sticky surfaces of pollen. Metals such as Cr, Co, Cu, Fe, Mn and Zn are essential for humans, and they may play an important role in a number of biochemical processes. Some of them are present at the trace level and being toxic if they exceed safety levels. Heavy metals pollution affects the quality of productions, as well as the qualities of the atmosphere and waters.
So, the presence of metals in honey may affect the health of human as a consumer. Analysis of the metals in honey helps its quality control as a food product [4,5, 6].
The metals are picked up by the roots and distributed throughout the entire plant including the nectar and pollen. Bees collect both the contaminated pollen and nectar and transport it back to the hive.
The aim of this study was to determine the concentrations of some heavy metals in some honey samples harvested from different locations in Romania using Atomic Absorbtion Spectrometer (AAS).
Romania has a very long tradition of beekeeping. All the samples were provided by the local association of bee keepers with guaranteed origin and made by traditional procedures in the honey- producing region and all honey samples were collected freshly in sterile containers (labeled with numbers, place and date of collection) and stored at low temperature (4-8 °C) until analyzed.
The results of such studies can help prevent the mentioned problems and improve the healthy honey consumption. These results can lead to considering the origins of honey contaminants such as soil type and air pollution, regarding the food safety in health policy, and providing best quality of food will protect public health and preserve consumer confidence.
2. Materials and Methods
The AAS measurements were performed using SHIMADZU AA 6300 (air/acetylene flame) spectrometer in order to determine the heavy metals concentration in honey samples: cadmium (λ =228.8 nm), copper (λ = 324.7 nm), zinc (λ = 213.9 nm), iron (λ = 248.3 nm) and lead (λ = 217 nm).
The objective of the current study was to determine the concentrations of some heavy metals like cadmium (Cd), copper (Cu), zinc (Zn), iron (Fe) and lead (Pb) in some honey samples (tilia honey, acacia honey, rapes honey and multiflora honey) harvested from different locations in Romania (Ploiesti, Campina, Fagaras).
The honey samples (fifteen different samples from each type of honey for each investigated zone) preparation for AAS measurements was following: honey sample was digested in an acid solution using a microwave digestion system and the clear solution volume was made up to 50 mL for each sample using deionized water.
1.0 g of each sample of honey was digested in a microwave digester with 5.0 mL concentrated 65% nitric acid and 1mL 30% hydrogen peroxide in PTFE vessels. The vessels were placed into microwave system (MARS 5, CEM).
A blank digest was carried out in the same way. Digestion conditions for the microwave system applied were as follows: up to 120 °C for 15 min and then constant for 10 min; up to 160 °C in 20 min and constant for 15 min; finally, a cooling stage (30 min) was carried out to 22 °C and diluted to 50 mL with deionized ultrapure water. The solutions were then transferred into a clean, dry plastic bottles awaiting analysis.
A blank solution was prepared by subjecting 2 mL of de-ionized water through the same process. All the samples were digested in triplicates.
Results are expressed as mean ± S.D (standard deviation) of triplicate analysis. The statistical calculations and analysis were performed using SPSS version 18.0. ANOVA was used for the analysis of the data obtained and the level of significance was taken up as p < 0.05.
All reagents were of analytical grade. Double-deionized water produced by Milli Q water purification system (Millipore) was used in all dilutions. The stock solutions of Cd, Cu, Zn, Fe and Pb (1000 mg/L, ICP standard CertiPUR) were purchased from Merck (Germany) and the elements standard solutions were prepared by diluting them. Honey samples were digested with concentrated nitric acid (65% HNO3, Sigma Aldrich, Germany) and hydrogen peroxide (30% H2O2 pure p.a, Sigma Aldrich, Germany). Heavy metal contents of samples were compared to standards and results were determined in ppm (μg/g).
3. Results and discussion
The heavy metal concentrations in the honey samples collected from Ploiesti has been presented in Table I.
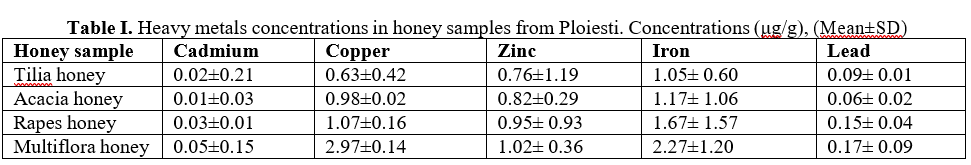
The highest concentrations of heavy metals were detected in multifloral and rapes honey samples. COMMISSION REGULATION (EU) 2015/1005 of 25 June 2015 amending Regulation (EC) No 1881/2006 as regards maximum levels of lead in certain foodstuffs. So, maximum levels of lead and cadmium in honey is 0.1 mg/kg [7].
In the rapes honey samples (0.15 μg/g) and multiflora honey samples (0.17 μg/g) the concentration maximum permitted of lead was exceeded. In urban areas, the industries release some gases containing lead as one of the contents. In the similar way, the fumes released from the gasoline combustion in vehicles also acts as a source of lead in environment which can be deposited in soil, water and plants and later enter humans through food chain.
Lead can contaminate air or nectar and honeydew. Presence of cadmium in honey samples is within the maximum permitted limit (0.01-0.05 μg/g). The Cd sources are: the usage of phosphate fertilizers, the presence in sewage sludge, smelting, or mining. Another contamination of honey may be through metal plating, pigments, and plastics with which entered in contact.
The analysis revealed the presence of Zinc (0.86-2.02 μg/g), Copper (0.63-3.97 μg/g) and Iron
(1.78-3.27 μg/g) in the collected samples.
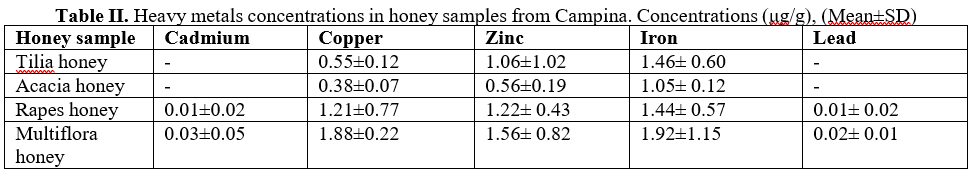
The heavy metal concentrations in the honey samples collected from Campina has been presented in Table II.
4. Not detected
The concentrations of cadmium and lead are very low, in tilia honey samples and acacia honey samples are under detected limits. The analysis revealed the presence of Zinc (0.56-1.56 μg/g), Copper (0.38-1.88 μg/g) and Iron (1.05-1.92 μg/g) in the collected samples, much lower than samples collected from Ploiesti.
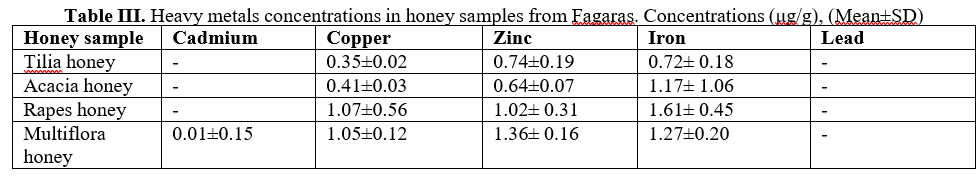
The heavy metal concentrations in the honey samples collected from Fagaras has been presented in Table III.
4. Not detected
The lead concentration was below detected limit, and cadmium concentration was almost in the same situation excepted multiflora honey samples. We remark from data presented in table III the lowest concentrations of metals in the honey samples collected from Fagaras: Zinc (0.64-1.36 μg/g), Copper (0.35-1.07 μg/g) and Iron (0.72-1.61 μg/g).
The amount of zinc in honey particularly depends on the instrument used in apiaries, centrifugation and storage of honey, transport utilities and technological process.
The copper can be presented in the honey due to the copper fungicides use into agriculture. The main man-made releases of copper are from coal-fired power stations, metal production, waste incinerators, sewage treatment processes, and from the application of agricultural chemicals. Smaller amounts are also released naturally from the earth’s crust.
Metal decomposition of iron utensils used for storage of honey in some sites is one of the basic reasons for presence of Fe in samples. The iron concentration in honey also depends on the flora of a particular area having a source of iron compounds.
5. Conclusions
Honey produced in different regions of Romania do not completely lack heavy metals but they are at satisfactory level and good quality for human consumption excepted the samples collected from Ploiesti, a zone with high industrial activity.
The analysis shows us that in the rapes honey samples (0.15 μg/g) and multiflora honey samples (0.17 μg/g) the concentration maximum permitted of lead was exceeded.
The above-obtained results make it possible to state that concentrations of heavy metals found in honey samples might be due to:
- the use of improper collecting container (made of various metallic alloys not recommended for preservation of honey);
- exhaust gas from vehicles (which use various fuels containing lead compounds);
- residues emanated or discharged by various industries.
Metals are released into the environment through their use in industrial processes and enter the food chain from uptake by plants from contaminated soil or water. Metals concentration in various places depends on many variables, leading to their different concentrations in honey. Some control measures like the quality control of food products, monitoring the soil in agricultural regions and limiting the use of fertilizers are recommended.
Contributo selezionato da Filodiritto tra quelli pubblicati nei Proceedings “17th Romanian National Congress of Pharmacy - 2018”
Per acquistare i Proceedings clicca qui:
https://www.filodiritto.com/proceedings
Contribution selected by Filodiritto among those published in the Proceedings “17th Romanian National Congress of Pharmacy - 2018”
To buy the Proceedings click here:



